The Closet Guardian! The Surprising Science Hidden in the “White Beads” of Moisture Absorbers (Calcium Chloride)
I am Ken Kuwako, your Science Trainer. Every day is an experiment!
During those humid seasons or when dealing with musty closets, “Dehumidifiers” are our best friends. Have you ever stopped to wonder about the “white granules” sitting inside that container? Believe it or not, these tiny grains are packed with amazing science that protects our homes. Today, let’s dive into the secret world behind your everyday dehumidifier.
The Unexpected Identity: Is This the Same Stuff from the Road?
 I recently bought a dehumidifier for my house. Glancing at the ingredients on the back, I saw the name Calcium Chloride. This name might ring a bell for some of you. That’s right—it is the exact same component found in the “anti-icing agents” or “road salt” scattered on snowy roads during freezing winters! It is quite a shock to realize that the stuff grabbing moisture in your closet is the same stuff melting ice on the highway.
I recently bought a dehumidifier for my house. Glancing at the ingredients on the back, I saw the name Calcium Chloride. This name might ring a bell for some of you. That’s right—it is the exact same component found in the “anti-icing agents” or “road salt” scattered on snowy roads during freezing winters! It is quite a shock to realize that the stuff grabbing moisture in your closet is the same stuff melting ice on the highway.
Self-Liquefying Magic: The Power of “Deliquescence”
Why is Calcium Chloride used as a dehumidifier? The secret lies in a unique property called Deliquescence. Here is how Wikipedia describes it:
The raw material is solid calcium chloride, utilizing its deliquescent property—where the substance absorbs moisture and turns into a liquid—for dehumidification. Its primary use is in closets, wardrobes, and shoe boxes, where it is most effective in sealed spaces. On the other hand, it has limited absorption capacity in large rooms and is ineffective due to frequent air exchange.
“Deliquescence” is a phenomenon where a substance pulls water vapor from the air so aggressively that it eventually dissolves in that water, turning itself into a liquid. When you see water pooling at the bottom of the container, it didn’t leak in from the outside. Instead, the white granules pulled moisture from the air and transformed into a liquid. Interestingly, when used on snowy roads, it utilizes its ability to release heat as it dissolves (heat of solution) and its ability to lower the freezing point of water. Same chemistry, different jobs!
Is This a Chemical Reaction?
When Calcium Chloride ($CaCl_2$) captures water vapor, it isn’t just “getting wet.” First, it pulls water molecules into its very own crystal structure, transforming into a different form of matter called a “hydrate.”
For example, when dry calcium chloride granules absorb water, the chemical equation looks like this:
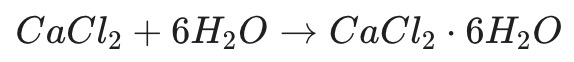
In this process, it stops being simple calcium chloride and becomes Calcium Chloride Hexahydrate.
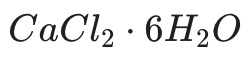
(Calcium Chloride Hexahydrate)
A “hydrate” is a substance where water molecules are incorporated into the crystal in a specific ratio. This “locking in” of water molecules can be considered a type of chemical reaction because the properties and structure change from the original dry granules.
As it continues to pull in more vapor, the hydrate eventually dissolves into the very water it captured, resulting in that familiar pool of liquid. This is the final stage of Deliquescence.
-
Combining with water molecules to become a “hydrate” (Strong chemical nature)
-
Dissolving into that water to become an “aqueous solution” (Strong physical nature)
It is a clever two-step process!
A Science Teacher’s Tip: Where to Use Your Dehumidifier
You can’t just throw a dehumidifier anywhere and expect miracles. As mentioned in the Wikipedia entry, it shows its true worth in sealed spaces. In a large, breezy living room, new moisture arrives constantly, and the calcium chloride simply can’t keep up.However, in “small, enclosed spaces” like closets or shoe racks, it effectively captures the moisture trapped inside. In a science classroom, calcium chloride is a fantastic tool for learning about states of matter and reactions. I am excited to see how much water my new dehumidifier “collects and transforms” in a month’s time.I encourage you all to look at your dehumidifiers with a scientific eye. It’s pretty cool to see physics and chemistry working right in your own home!
Inquiries and Requests
Making the wonders of science more accessible! I share easy and fun science experiments you can do at home, along with tips and tricks. Feel free to explore!・The “Science Notes” content is now a book! Details here.・About the operator, Ken Kuwako, here.・For various requests (writing, lectures, workshops, TV supervision/appearance), click here.・Updates are available on X (Twitter)!
![]() Check out experiment videos on the Science Material Channel!
Check out experiment videos on the Science Material Channel!
5月のイチオシ実験!
キーンと冷えるドライアイス!気温が上がってくるこの時期・ドライアイスを使った昇華・凝結・等速度直線運動の実験はいかが?

液体ゼロ!ドライアイスが消えるまでの3時間を科学する(昇華・凝結・等速度直線運動)
テレビ番組監修・イベント等のお知らせ
- 4月30日(木)「THE突破ファイル」(日本テレビ)の科学監修を担当しました。
- 5月8日(金)理科教育ニュースを担当しました。
- 6月14日(日) 千葉大学インスタレーション「探究」にて講師を務めます
- 6月26日(金) 千葉大学の公開研究会(中学理科について授業公開予定)
- 7月18日(土) 教員向け実験講習会「ナリカカサイエンスアカデミー」の講師をします。お会いしましょう。
書籍のお知らせ
- 『大人のための高校物理復習帳』(講談社)…一般向けに日常の物理について公式を元に紐解きました。特設サイトでは実験を多数紹介しています。※増刷がかかり6刷となりました(2026/02/01)

- 『きめる!共通テスト 物理基礎 改訂版』(学研)… 高校物理の参考書です。イラストを多くしてイメージが持てるように描きました。授業についていけない、物理が苦手、そんな生徒におすすめです。特設サイトはこちら。

各種SNS(更新情報をお届け!)
X(Twitter)/instagram/Facebook(日本語)
Explore
- 楽しい実験…お子さんと一緒に夢中になれるイチオシの科学実験を多数紹介しています。また、高校物理の理解を深めるための動画教材も用意しました。
- 理科の教材… 理科教師をバックアップ!授業の質を高め、準備を効率化するための選りすぐりの教材を紹介しています。
- Youtube…科学実験等の動画を配信しています。
- 科学ラジオ …科学トピックをほぼ毎日配信中!AI技術を駆使して作成した「耳で楽しむ科学」をお届けします。
- 講演 …全国各地で実験講習会・サイエンスショー等を行っています。
- About …「科学のネタ帳」のコンセプトや、運営者である桑子研のプロフィール・想いをまとめています。
- お問い合わせ …実験教室のご依頼、執筆・講演の相談、科学監修等はこちらのフォームからお寄せください。