A Science Show Smash Hit! Find the Perfect Formula for Elephant Toothpaste!
Hi, I’m Ken Kuwako, your Science Trainer. Every day is an experiment!
【This article is also available on my radio show!】
Have you ever heard of the experiment called Elephant’s Toothpaste? Once the reaction starts, a massive, fluffy fountain of foam bursts out, looking just like a giant tube of toothpaste being squeezed! It’s a high-impact experiment that’s a huge hit in science classrooms and live shows. Recently, I teamed up with my students to give it a try. Check out the video below to see the explosive results!
To pull off a successful science show, I spent some time figuring out the perfect “recipe”—one that produces maximum foam while staying safe. Hydrogen peroxide is constantly breaking down into oxygen and water, which is why we keep it in the fridge. However, without a little help, this reaction is incredibly slow. By adding potassium iodide to the hydrogen peroxide, we trigger a rapid release of oxygen, creating a mountain of bubbles in an instant. The potassium iodide acts as a catalyst, kickstarting the decomposition process all at once.
Here is the chemical equation for the reaction:

The size and intensity of the foam depend on the concentration and amount of hydrogen peroxide used. Higher concentrations lead to faster, more dramatic reactions. However, if the reaction is too violent, it can be hard to control—finding that perfect balance is the tricky part! We always conduct these tests with safety as our top priority. It’s also worth noting that this is an exothermic reaction; if you touch the flask or the foam afterward, you’ll feel a distinct warmth. Beyond the visual spectacle, it’s a fantastic way to learn about reaction heat and catalysts.
Experimental Methods
I tested three different variations to see how the results changed.
1. 50mL Hydrogen Peroxide + 3 tbsp Potassium Iodide (dissolved in 50mL warm water)
First, I poured 50mL of hydrogen peroxide into a 500mL Erlenmeyer flask, adding a dash of dish soap and some food coloring for effect. I placed the flask inside a plastic bin and then added the potassium iodide solution (3 tablespoons dissolved in water).

The moment the potassium iodide hits the liquid, oxygen is released so rapidly that the dish soap captures it into a massive structure of bubbles.


With 50mL, the foam filled up the bin nicely. The final volume of foam is directly tied to how much hydrogen peroxide you start with.


Reference: Using Solid Potassium Iodide
When I added the potassium iodide as a solid powder instead of a solution, the reaction looked more like “snake fireworks.” It was much slower, allowing us to observe a long, steady crawl of foam.

A word of caution: if you do this without dish soap, the reaction is violent and the solution can splash everywhere. I also tried a mini-version using just 5mL of hydrogen peroxide in a graduated cylinder. After the reaction, I lowered a glowing wooden splint into the cylinder, and it burst back into flames—proving that the gas being produced was indeed pure oxygen!

2. 200mL Hydrogen Peroxide + 6 tbsp Potassium Iodide (dissolved in 50mL warm water)
Next, I scaled things up. 200mL of hydrogen peroxide paired with a solution of 6 tablespoons of potassium iodide. Here is what happened:

200mL Hydrogen Peroxide
The foam overflowed and covered the entire desk!

This felt like the “Golden Ratio” for a great visual, though I think I used slightly too much water to dissolve the potassium iodide.
3. 200mL Hydrogen Peroxide + 6 tbsp Potassium Iodide (dissolved in 30mL warm water)
Finally, I tried the same 200mL of hydrogen peroxide but concentrated the catalyst by using only 30mL of warm water for the potassium iodide.

Another desk-engulfing success!
For those curious, the dish soap I used was “Kyukyutto” (a popular Japanese brand).

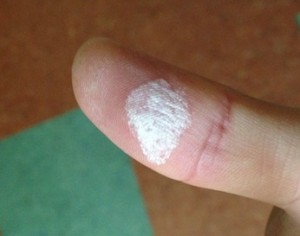
A very important safety note: while weak hydrogen peroxide is sold as a common antiseptic (Oxydol), high-concentration hydrogen peroxide is dangerous. It is corrosive and can cause chemical burns. Even the foam looks tempting to touch, but please don’t! It will turn your skin white and cause a painful stinging sensation that lasts all day. Always wear gloves, a lab coat, and safety goggles. Cleanup should always be handled by the instructor. I actually accidentally touched some during the cleanup, and my skin turned white immediately. Stay safe and keep your distance from the foam!
This is what happens when you touch it—my own skin after an accidental “human experiment.” The stinging lasted for quite a while. Click here for the explanatory slides created by our Science Club! It’s a great resource.
Contact & More
Let’s make the wonders of science a part of your daily life! I’ve put together plenty of fun experiments and tips you can try at home. Feel free to explore!
・My book “Science Notebook” is now available. Details here.
・Learn more about me, Ken Kuwako, here.
・For inquiries (writing, speaking, workshops, TV supervision, etc.), click here.
・Get the latest updates on X (formerly Twitter)!
![]() Check out my experiment videos on the Science Material Channel!
Check out my experiment videos on the Science Material Channel!
5月のイチオシ実験!
キーンと冷えるドライアイス!気温が上がってくるこの時期・ドライアイスを使った昇華・凝結・等速度直線運動の実験はいかが?

液体ゼロ!ドライアイスが消えるまでの3時間を科学する(昇華・凝結・等速度直線運動)
テレビ番組監修・イベント等のお知らせ
- 4月30日(木)「THE突破ファイル」(日本テレビ)の科学監修を担当しました。
- 5月8日(金)理科教育ニュースを担当しました。
- 6月14日(日) 千葉大学インスタレーション「探究」にて講師を務めます
- 6月26日(金) 千葉大学の公開研究会(中学理科について授業公開予定)
- 7月18日(土) 教員向け実験講習会「ナリカカサイエンスアカデミー」の講師をします。お会いしましょう。
書籍のお知らせ
- 『大人のための高校物理復習帳』(講談社)…一般向けに日常の物理について公式を元に紐解きました。特設サイトでは実験を多数紹介しています。※増刷がかかり6刷となりました(2026/02/01)

- 『きめる!共通テスト 物理基礎 改訂版』(学研)… 高校物理の参考書です。イラストを多くしてイメージが持てるように描きました。授業についていけない、物理が苦手、そんな生徒におすすめです。特設サイトはこちら。

各種SNS(更新情報をお届け!)
X(Twitter)/instagram/Facebook(日本語)
Explore
- 楽しい実験…お子さんと一緒に夢中になれるイチオシの科学実験を多数紹介しています。また、高校物理の理解を深めるための動画教材も用意しました。
- 理科の教材… 理科教師をバックアップ!授業の質を高め、準備を効率化するための選りすぐりの教材を紹介しています。
- Youtube…科学実験等の動画を配信しています。
- 科学ラジオ …科学トピックをほぼ毎日配信中!AI技術を駆使して作成した「耳で楽しむ科学」をお届けします。
- 講演 …全国各地で実験講習会・サイエンスショー等を行っています。
- About …「科学のネタ帳」のコンセプトや、運営者である桑子研のプロフィール・想いをまとめています。
- お問い合わせ …実験教室のご依頼、執筆・講演の相談、科学監修等はこちらのフォームからお寄せください。